Reading time
5
min read
Date
Written by
Rich Roginski (Founder)

The FDA Just Changed a 60-Year Standard. Here's What Biotech Needs to Know
The FDA just ended a 60-year standard. One trial is now the default. For biotech teams, that means every recruitment decision, every regulatory submission, and every site selection call carries more weight than ever before. FutureNova Health breaks down what changed, what it means for your pipeline, and why precision-first execution is no longer optional.

Next-level precision in clinical trial recruitment
Today, the FDA officially abandoned its decades-old requirement of two clinical trials for new drug approvals.
FDA Commissioner Dr. Marty Makary and deputy Dr. Vinay Prasad published the announcement in the New England Journal of Medicine, stating the agency's new default position will be one pivotal study for new drugs and novel health products. A standard in place since 1962 is now officially behind us.
Their reasoning is straightforward: drug development has become precise enough that a single well-designed trial, supported by biomarker data, mechanistic evidence, and related findings, can tell a complete story. As they wrote, "overreliance on two trials no longer makes sense" in 2026.
What this actually means for your pipeline:
For oncology and rare disease sponsors, this is largely a formalization of what was already happening. Roughly 60% of first-of-a-kind approvals over the last five years were already based on a single study. The bigger impact lands on common disease indications, where two trials had remained the norm.
One trial is now enough to earn approval. That is the opportunity. But it only works in your favor if your execution is precise from day one. Patient targeting. Site selection. Regulatory submissions. Enrollment velocity. The sponsors who win in this environment will be the ones who treat every decision as a precision call, not the ones counting on a second trial to fill gaps the first one left behind.
Precision isn't optional anymore. It's the whole game.
This is where AI-driven clinical strategy changes outcomes. Our NovaNext platform was built for exactly this kind of environment:
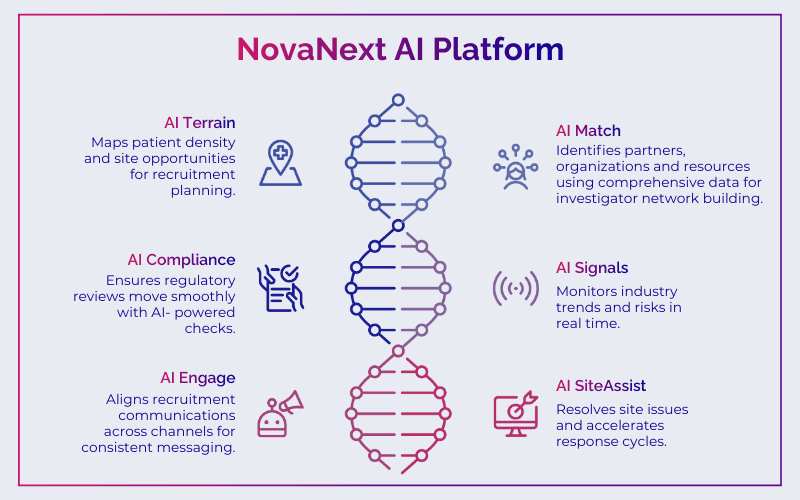
NovaNext AI Terrain maps geographic patient density and site opportunity before you commit resources, so recruitment planning starts with clarity, not assumptions.
NovaNext AI Match identifies the right KOLs for your indication using comprehensive data, not incomplete manual research, so your investigator network is built on precision from day one.
NovaNext AI Compliance keeps regulatory reviews moving with AI-powered checks that catch risk before it stalls your submission. Precise. Thorough. Faster.
NovaNext AI Signals monitors the industry landscape in real time so your team never misses a competitive signal, a site performance flag, or a protocol risk hiding in the noise.
NovaNext AI Engage aligns your recruitment communications across every channel and touchpoint, so your message reaches the right participants consistently and compliantly.
NovaNext AI Site Assist resolves routine site issues and accelerates response cycles, keeping your sites focused on enrollment rather than administrative friction.
The sponsors who win in this new environment won't be the ones with the biggest budgets. They'll be the ones with the sharpest execution.
If this week's announcement has you rethinking your recruitment and regulatory strategy, that's exactly the right instinct.
Let's talk about what precision-first clinical recruitment looks like for your next trial.
One trial changes everything about how you plan, recruit, and execute. Reach out and ask about our risk-free Test Flight program. Experience what AI-driven precision looks like for your trial before you commit to anything.