Reading time
7
min read
Date
Written by
Rich Roginski (Founder)

AI in 2026: What Healthcare Marketers Should Do After 2025’s Experiments
Lessons we learned in the AI world in 2025 and what this year is looking like for Clinical Trial Recruitment 1 month into 2026.

What Changed: AI in Clinical Trial Recruitment (2025 Lessons)
The problem isn't new. 80% of clinical trials miss enrollment deadlines. 37-48% of activated sites under-enroll or fail to recruit a single patient. Each recruitment delay costs your trial roughly $40,000 per day in direct costs, with Phase III daily costs exceeding $55,000.
What changed in 2025 is how AI exposed the gap between companies who built digital-first recruitment infrastructure and those still retrofitting legacy workflows.
How Patient Discovery Shifted in 2025
Patients stopped starting their trial journey on sponsor websites. They now begin with AI-powered search experiences, conversational tools, and chat-based discovery that surface answers before they ever click a link (Definitive Healthcare, Healthcare Success).
Your trial becomes invisible if your content isn't structured for AI retrieval. Geographic targeting, eligibility criteria, and patient education materials must now feed AI systems that increasingly control top-of-funnel discovery.
Traditional recruitment vendors caught flat-footed. Digital-first teams gained months.
Where AI Actually Worked
Three applications proved reliable enough to scale in 2025:
AI-Assisted Pre-Screening and Geo-Targeting NovaNext's geo-targeting tools now use real-world data and predictive analytics to identify high-probability patient populations by location, eliminating wasted ad spend on low-yield markets. Sites receive qualified leads instead of noise.
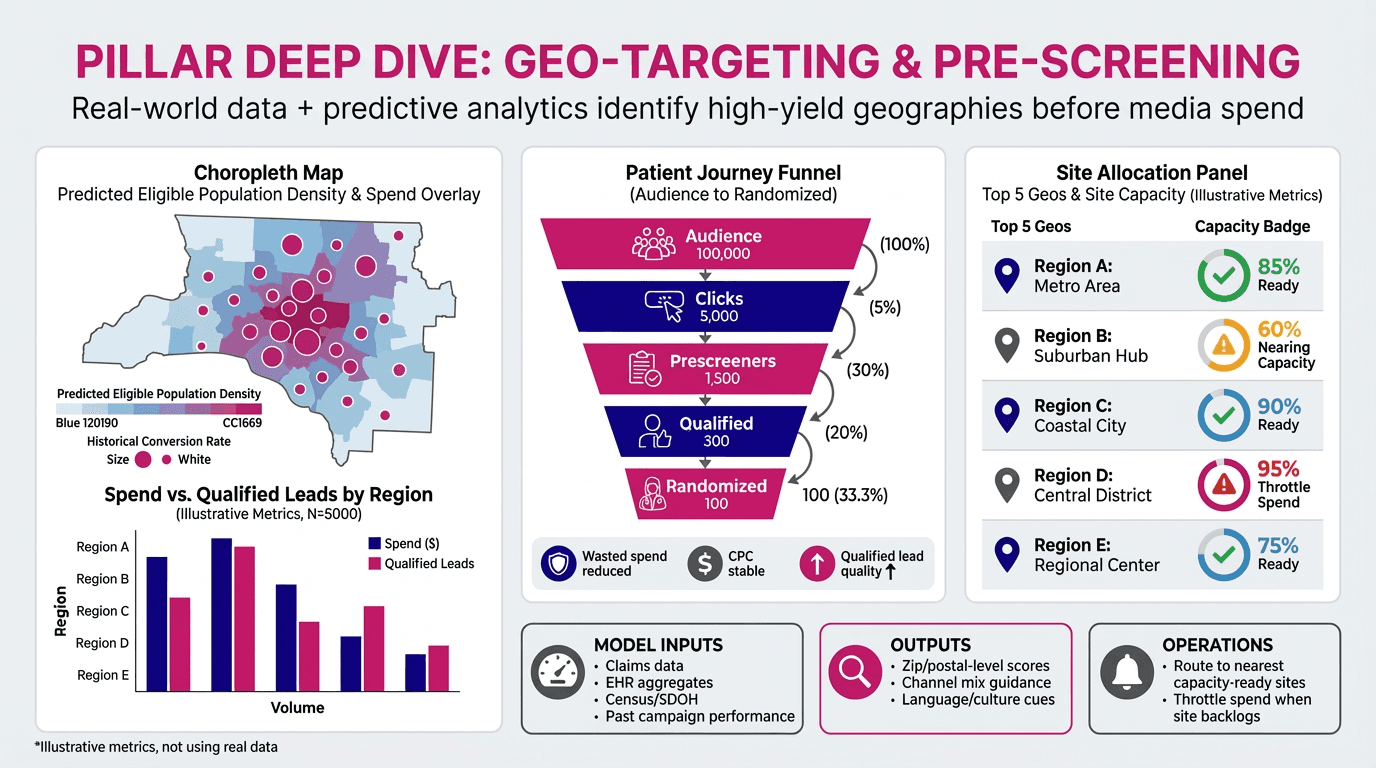
Personalized Patient Education at Scale Long-form consent documents and protocol summaries were transformed into micro-content: short explainer videos, interactive FAQ modules, SMS journeys triggered by engagement behavior. AI repurposed approved materials without inventing new claims (ON24, Healthcare Success).
This shift moved recruitment from one-time campaigns to always-on education that adapts to each participant's questions and concerns.
Operational Intelligence for Site Performance AI dashboards surfaced patterns traditional CROs miss: which sites consistently under-perform, which messaging drives conversion, which eligibility barriers cause the most drop-offs. Trial teams could intervene weeks earlier instead of discovering problems during quarterly reviews.
What Didn't Work
AI that improvised science or generated unapproved claims. Teams that tried to bolt AI onto paper-based workflows without changing underlying processes. Recruitment strategies that ignored caregiver decision-makers in rare disease trials or failed to address geographic and cultural barriers.
Compliance remains non-negotiable. AI must re-express approved content, not invent new efficacy language.
The 2026 Reality
Recruitment is no longer just a site‑selection and advertising problem; it is increasingly a digital infrastructure problem shaped by regulatory, operational, and technological constraints.
FDA's FDORA‑enabled Diversity Action Plan framework is pushing sponsors toward deliberate, documented diversity strategies, even though the requirement is not yet mandatory while final guidance remains pending. Parallel pressures are pushing trials toward more decentralized‑trial elements, because fixed‑site models struggle to improve access and reduce patient burden when protocol complexity and logistical demands keep rising. Analyses of protocol amendments show that 57% of Phase II–III protocols face at least one substantial amendment, with median direct‑implementation costs of approximately $141,000 for Phase II and $535,000 for Phase III, and these changes often extend timelines by about 3 unplanned months per substantial amendment.
AI‑driven tools alone will not resolve these structural issues, but digital‑first recruitment infrastructure—built around structured protocols, eligibility data, and automated screening—can shorten the time between protocol approval and first patient enrolled, reduce per‑patient acquisition costs, and provide real‑time visibility into which recruitment strategies are actually working. Generative AI applied to clinical‑trial eligibility screening (e.g., GPT‑4 applied to EHR data) demonstrates 97–100% accuracy in identifying eligible patients at ~11 cents per patient, compared to manual screening at tens of thousands of dollars annually and 91–100% accuracy by human study staff. This evidence supports the case that intelligent infrastructure—not just more ads—can compress enrollment timelines and reduce costs without sacrificing quality or oversight.
Ever-Evolving AI-Structured Discovery (Not Just Traditional SEO)
Trial content must now be structured for AI-powered discovery, not just traditional search engines.
Eligibility criteria, geographic targeting, and protocol summaries need clean, machine-readable formatting to surface in AI-driven answer engines.
Patient education materials must be modular and structured so AI systems can retrieve approved language without improvisation.
Metadata, structured data, and compliance-approved content blocks now influence discoverability upstream of sponsor websites.
Digital-first recruitment teams are building content ecosystems designed for AI retrieval, not retrofitting static PDFs after the fact.
How FutureNova Built for This
We built digital-first from day one. Beyond the AI SEO structure above, we built digital-first from day one. We didn't retrofit.
NovaNext Platform: Purpose-built AI tools for geo-targeting, signals intelligence, KOL identification, and compliance-checked content generation. FDA integration ensures every piece of patient-facing material stays within approved boundaries.
Navigator Model: Your clinical recruitment lead isn't an account manager passing requests to vendors. They're an industry veteran who executes strategy, creative, and media directly. One point of contact. One source of truth. Zero unnecessary layers.
Trial-Ready Digital Portals: HIPAA-compliant, mobile-optimized participant access that works across devices and supports decentralized trial requirements without custom development.
If 2025 proved anything, it's that recruitment delays are existential business risks, not operational nuisances. Each day of delay is $40,000 in direct costs and up to $500,000 in lost revenue.
The teams who solved recruitment in 2025 weren't running better ads. They built better infrastructure.
Ready to eliminate recruitment as your biggest delay risk? Let's talk about how NovaNext cuts your timeline without compromising compliance. Reach out today!
Sources:
Tufts CSDD 2024 analysis, Definitive Healthcare 2026 outlook, Healthcare Success 2025 research, ON24 life sciences report, FDA FDORA guidance Varma, T., Bierer, B. E., & Hantel, A. (2024). "The Promise and Perils of Diversity Action Plans for Clinical Trials." JAMA, 332(21), 1787–1788. PMC., "Understanding the gap between expectations and reality in..." PMC. 2025. Getz, K. A., et al. (2016). "The Impact of Protocol Amendments on Clinical Trial Performance and Cost." Therapeutic Innovation & Regulatory Science, 50(4), 436–441. Tufts CSDD / PubMed., Mass General Brigham / NEJM AI. (2024). "Generative AI for Clinical Trial Eligibility Screening." Study demonstrates 97–100% accuracy at ~11 cents per patient. New England Journal of Medicine (AI edition)., "Opportunities and counterintuitive challenges for decentralized..." Nature. 2022., Crowell & Moring. (2026). "After Trump Executive Orders, FDA Removes Diversity Guidance from Website.", FDA. (2025). "Diversity Action Plans to Improve Enrollment of Participants." FDA guidance documents and compliance summary.